Electroforming and electroplating are two common metal forming processes that use electricity to deposit metal onto a surface. Both processes involve passing an electric current through a solution called an electrolyte, which contains metal ions. The metal ions are attracted to the surface of an object called an electrode, where they form a solid metal layer. The electrode can be either a cathode, which is the negative terminal of the electric circuit, or an anode, which is the positive terminal of the electric circuit.
Electroforming and electroplating have different purposes, methods, and outcomes. Electroforming is a process of creating a metal part from scratch by depositing metal onto a non-metallic or sacrificial mold. Electroplating is a process of coating a metal part with another metal to improve its appearance, functionality, or durability. Electroforming and electroplating also have different characteristics, such as the thickness, quality, and composition of the metal layer. Electroforming produces a thick, high-quality, and homogeneous metal layer, while electroplating produces a thin, low-quality, and heterogeneous metal layer.
Electroforming and electroplating have a long history and a wide range of applications. Electroforming was invented in the 1830s by Moritz von Jacobi, a German-Russian physicist and engineer. Electroplating was invented in the 1800s by Luigi Brugnatelli, an Italian chemist and physicist. Electroforming and electroplating are used in various industries, such as jewelry, art, medical, and aerospace, to create unique and complex metal parts or to improve the performance and durability of metal parts.
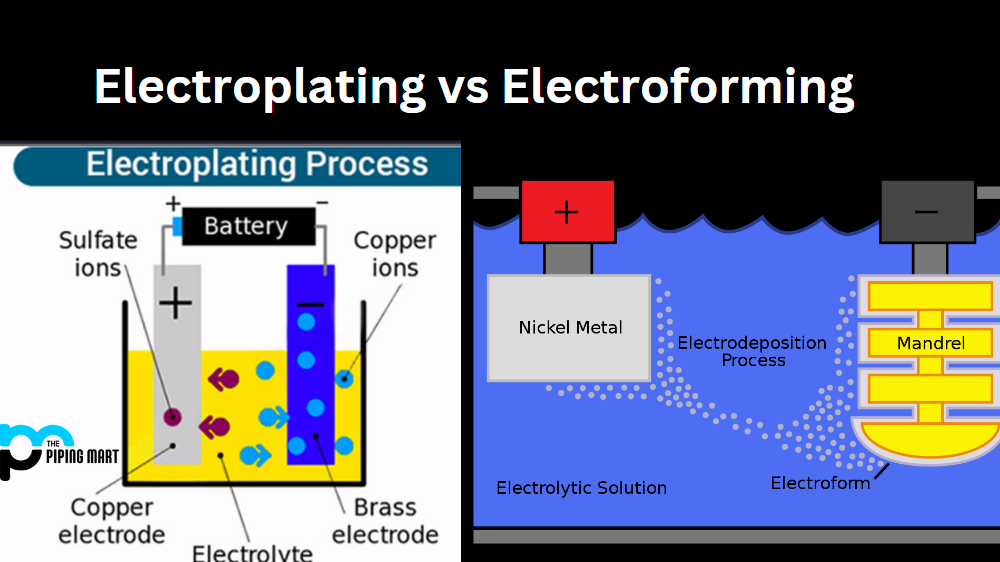
The electroforming process is the process of creating a metal part from scratch by depositing metal onto a non-metallic or sacrificial mold. The mold can be made of various materials, such as plastic, wax, or resin, and can have any shape or size. The mold is also coated with a conductive layer, such as silver or graphite, to allow the electric current to flow through it. The mold is then placed in an electrolyte solution, which contains metal ions of the desired metal, such as copper, nickel, or gold. The mold is connected to the negative terminal of the electric circuit, making it the cathode. The metal ions are attracted to the cathode, where they form a solid metal layer. The metal layer grows thicker and thicker, until it reaches the desired thickness or covers the entire mold. The mold is then removed from the electrolyte solution, and the metal part is separated from the mold. The mold can be either dissolved, melted, or peeled off, depending on the material and the shape of the part.
Steps of the Electroforming Process
The mold is prepared by creating the desired shape and size, and coating it with a conductive layer. The mold can be created by using various methods, such as 3D printing, CNC machining, or casting. The conductive layer can be applied by using various methods, such as spraying, painting, or sputtering.
The electrolyte is prepared by dissolving the metal salts or metal anodes in a solvent, such as water or organic solvents. The electrolyte can also contain additives, such as brighteners, levelers, or inhibitors, to improve the quality and appearance of the metal deposit. The electrolyte can also be heated, stirred, or filtered, to maintain its stability and purity.
The electroforming setup consists of a power supply, an anode, a cathode, and a container. The power supply provides the electric current and voltage for the electroforming process. The anode is the positive electrode that supplies the metal ions to the electrolyte. The anode can be either a metal plate or a metal rod, depending on the shape and size of the part. The cathode is the negative electrode that receives the metal ions from the electrolyte. The cathode is the mold that is coated with a conductive layer. The container is the vessel that holds the electrolyte solution and the electrodes. The container can be made of various materials, such as glass, plastic, or metal, depending on the type and temperature of the electrolyte.
The electroforming operation is the process of depositing metal onto the mold by passing an electric current through the electrolyte. The electric current and voltage can be adjusted to control the rate and quality of the metal deposit. The electroforming time can vary from minutes to hours, depending on the thickness and complexity of the part. The electroforming temperature can range from room temperature to 80 °C, depending on the type and concentration of the electrolyte. The electroforming process can also be monitored and controlled by using various instruments, such as ammeters, voltmeters, thermometers, or pH meters, to ensure the optimal conditions and results.
The part removal is the process of separating the metal part from the mold after the electroforming process is completed. The part removal can be done by using various methods, such as dissolving, melting, or peeling off the mold, depending on the material and the shape of the part. The part removal can also involve cleaning, polishing, or finishing the metal part, to remove any residues, defects, or rough edges.
The electroforming process can be affected by various factors, such as the mold material, the conductive layer, the electrolyte composition, the electric current and voltage, the electroforming time and temperature, and the part removal method. These factors can influence the quality and thickness of the metal deposit, as well as the shape and size of the part. Therefore, it is important to optimize and control these factors to achieve the desired outcome.

The electrophoretic painting process is the process of coating a metal part with another metal to improve its appearance, functionality, or durability. The metal part can be made of various metals, such as steel, copper, or brass, and can have any shape or size. The metal part is also cleaned and activated to ensure a good adhesion of the metal coating. The metal part is then placed in an electrolyte solution, which contains metal ions of the desired metal, such as silver, gold, or chrome. The metal part is connected to the negative terminal of the electric circuit, making it the cathode. The metal ions are attracted to the cathode, where they form a solid metal coating. The metal coating grows thinner and thinner, until it reaches the desired thickness or covers the entire part. The part is then removed from the electrolyte solution, and the metal coating is dried and polished to achieve the desired finish.
The process of electroplating process
The part is prepared by cleaning and activating it to remove any dirt, grease, oil, or oxide film that may interfere with the electroplating reaction. The cleaning can be done by using alkaline or acidic cleaners, solvents, or abrasives. The activating can be done by using acids, bases, or salts, to create a rough and reactive surface.
The electrolyte is prepared by dissolving the metal salts or metal anodes in a solvent, such as water or organic solvents. The electrolyte can also contain additives, such as brighteners, levelers, or inhibitors, to improve the quality and appearance of the metal coating. The electrolyte can also be heated, stirred, or filtered, to maintain its stability and purity.
The electroplating setup consists of a power supply, an anode, a cathode, and a container. The power supply provides the electric current and voltage for the electroplating process. The anode is the positive electrode that supplies the metal ions to the electrolyte. The anode can be either a metal plate or a metal rod, depending on the shape and size of the part. The cathode is the negative electrode that receives the metal ions from the electrolyte. The cathode is the metal part that is coated with another metal. The container is the vessel that holds the electrolyte solution and the electrodes. The container can be made of various materials, such as glass, plastic, or metal, depending on the type and temperature of the electrolyte.
The electrophoresis plating operation is the process of coating the metal part with another metal by passing an electric current through the electrolyte. The electric current and voltage can be adjusted to control the rate and quality of the metal coating. The electroplating time can vary from seconds to minutes, depending on the thickness and complexity of the part. The electroplating temperature can range from room temperature to 60 °C, depending on the type and concentration of the electrolyte. The electroplating process can also be monitored and controlled by using various instruments, such as ammeters, voltmeters, thermometers, or pH meters, to ensure the optimal conditions and results.
The part removal is the process of separating the metal part from the electrolyte after the electroplating process is completed. The part removal can be done by rinsing, drying, and polishing the metal part, to remove any residues, defects, or rough edges.
The electrophoretic coating process can be affected by various factors, such as the part material, the electrolyte composition, the electric current and voltage, the electroplating time and temperature, and the part removal method. These factors can influence the quality and thickness of the metal coating, as well as the shape and size of the part. Therefore, it is important to optimize and control these factors to achieve the desired outcome.

Electroforming materials are the types of materials that can be used for electroforming, such as metals, alloys, and composites. Electroforming materials have different characteristics, such as the composition, density, hardness, conductivity, and color, that affect the quality and performance of the electroformed parts. Electroforming materials can be classified into two categories: the substrate materials and the deposit materials.
Substrate materials are the materials that form the mold or the base for the electroforming process. Substrate materials can be made of various non-metallic or sacrificial materials, such as plastic, wax, or resin. Substrate materials can have any shape or size, depending on the desired shape and size of the electroformed part. Substrate materials can also be coated with a conductive layer, such as silver or graphite, to allow the electric current to flow through them. Substrate materials can influence the quality and performance of the electroformed part, such as the adhesion, accuracy, and smoothness. Therefore, it is important to select and prepare the substrate materials according to the specific application and requirements of the electroformed part.
Deposit materials are the materials that form the metal layer or the final product of the electroforming process. Deposit materials can be made of various metals, alloys, or composites, such as copper, nickel, or gold. Deposit materials can have different characteristics, such as the composition, density, hardness, conductivity, and color, that affect the quality and performance of the electroformed part. Deposit materials can also be mixed with other elements, such as carbon, silicon, or boron, to create new or improved properties, such as strength, wear resistance, or corrosion resistance. Deposit materials can influence the quality and performance of the electroformed part, such as the thickness, uniformity, and appearance. Therefore, it is important to select and control the deposit materials according to the specific application and requirements of the electroformed part.
Electroforming materials can be used to create a wide range of metal parts with different shapes, sizes, and complexities, such as jewelry, art, medical, and aerospace parts. Electroforming materials can also be used to create metal parts that are difficult or impossible to produce by other metal forming processes, such as casting, forging, or machining.
Electroforming materials can produce high-quality metal parts with high accuracy, precision, and resolution. Electroforming materials can also produce metal parts with high strength, hardness, and durability, as well as high optical, electrical, and thermal properties.
Electroforming materials can reduce the cost of metal parts production, as they require less material, energy, and labor. Electroforming materials can also reduce the waste and environmental impact of metal parts production, as they produce less scrap, emissions, and effluents.
Electroforming materials have some limitations in terms of the type and quality of the material. Electroforming materials can only produce metal parts that are made of the same material as the deposit material, which limits the choice and diversity of the material. Electroforming materials can also produce metal parts that have some defects, such as porosity, cracks, or stress, which can affect the performance and reliability of the part.
Electroforming materials have some challenges in terms of the optimization and control of the material. Electroforming materials require careful selection and preparation of the substrate and deposit materials, as well as the optimization and control of the electroforming process parameters, such as the electric current, voltage, time, and temperature. Electroforming materials also require proper testing and verification of the material properties and performance, as well as the quality and compliance of the part.

Electroplating materials are the types of materials that can be used for electroplating, such as metals, alloys, and composites. Electroplating materials have different characteristics, such as the composition, density, hardness, conductivity, and color, that affect the quality and performance of the electroplated parts. Electroplating materials can be classified into two categories: the base materials and the coating materials.
Base materials are the materials that form the part or the substrate for the electroplating process. Base materials can be made of various metals, such as steel, copper, or brass. Base materials can have any shape or size, depending on the desired shape and size of the electroplated part. Base materials can also be cleaned and activated to ensure a good adhesion of the metal coating. Base materials can influence the quality and performance of the electroplated part, such as the corrosion resistance, electrical conductivity, and mechanical strength. Therefore, it is important to select and prepare the base materials according to the specific application and requirements of the electroplated part.
Coating materials are the materials that form the metal layer or the final product of the electroplating process. Coating materials can be made of various metals, alloys, or composites, such as silver, gold, or chrome. Coating materials can have different characteristics, such as the composition, density, hardness, conductivity, and color, that affect the quality and performance of the electroplated part. Coating materials can also be mixed with other elements, such as zinc, nickel, or tin, to create new or improved properties, such as wear resistance, solderability, or lubricity. Coating materials can influence the quality and performance of the electroplated part, such as the thickness, uniformity, and appearance. Therefore, it is important to select and control the coating materials according to the specific application and requirements of the electroplated part.
Electroplating materials can improve the functionality of the metal parts by enhancing their properties or performance. Electroplating materials can provide corrosion resistance, electrical conductivity, thermal conductivity, or magnetic properties, depending on the type and composition of the coating material. Electroplating materials can also provide wear resistance, solderability, or lubricity, depending on the type and composition of the coating material.
Electroplating materials can improve the appearance of the metal parts by changing their color or finish. Electroplating materials can provide various colors, such as silver, gold, or chrome, depending on the type and composition of the coating material. Electroplating materials can also provide various finishes, such as matte, glossy, or textured, depending on the type and composition of the coating material.
Electroplating materials can improve the durability of the metal parts by increasing their lifespan or reducing their maintenance. Electroplating materials can protect the metal parts from corrosion, wear, or damage, depending on the type and thickness of the coating material. Electroplating materials can also reduce the need for cleaning, polishing, or repairing the metal parts, depending on the type and quality of the coating material.
Electroplating materials have some limitations in terms of the type and quality of the material. Electroplating materials can only produce metal parts that are coated with another metal, which limits the choice and diversity of the material. Electroplating materials can also produce metal parts that have some defects, such as porosity, cracks, or stress, which can affect the performance and reliability of the part.
Electroplating materials have some challenges in terms of the optimization and control of the material. Electroplating materials require careful selection and preparation of the base and coating materials, as well as the optimization and control of the electroplating process parameters, such as the electric current, voltage, time, and temperature. Electroplating materials also require proper testing and verification of the material properties and performance, as well as the quality and compliance of the part.
In this article, we have discussed the electroforming process, the electroplating process, the electroforming materials, the electroplating materials, the electroforming cost, the electroplating cost, the electroforming applications, and the electroplating applications. We have learned that electroforming and electroplating are both metal forming processes that use electricity to deposit metal onto a surface. However, they have different purposes, methods, and outcomes. Electroforming is a process of creating a metal part from scratch by depositing metal onto a non-metallic or sacrificial mold. Electroplating is a process of coating a metal part with another metal to improve its appearance, functionality, or durability. Electroforming and electroplating also have different characteristics, such as the thickness, quality, and composition of the metal layer. Electroforming produces a thick, high-quality, and homogeneous metal layer, while electroplating produces a thin, low-quality, and heterogeneous metal layer.
Electroforming and electroplating have various advantages and disadvantages, depending on the type and quality of the material. Electroforming and electroplating can produce versatile, high-quality, and low-cost metal parts, but they also have some limitations, challenges, and environmental and health impacts. Therefore, it is important to understand the differences and similarities between electroforming and electroplating, and to select and control the appropriate materials and processes for the specific application and requirements of the metal parts.
 Ferrous vs Non-Ferrous Metals: A Comparison of Properties and ApplicationsDecember 4, 2023Metals are one of the most important materials in human history. They have been used for various purposes, such as tools, weapons, coins, jewelry, art, and construction. Metals can be classified into two main categories: ferrous and non-ferrous metals.view
Ferrous vs Non-Ferrous Metals: A Comparison of Properties and ApplicationsDecember 4, 2023Metals are one of the most important materials in human history. They have been used for various purposes, such as tools, weapons, coins, jewelry, art, and construction. Metals can be classified into two main categories: ferrous and non-ferrous metals.view What Kind of Parts Are 5-Axis Suitable for Machining?October 30, 2023Over the past few years, CNC machining technology has evolved from simple machines to more complex ones. 5-axis CNC machining is one of the most advanced machining processes available today. It utilizes subtractive machining techniques that offer endless possibilities for part creation. The technology uses cutting tools that work on 5 axes to cut the workpiece into the desired shape and size.view
What Kind of Parts Are 5-Axis Suitable for Machining?October 30, 2023Over the past few years, CNC machining technology has evolved from simple machines to more complex ones. 5-axis CNC machining is one of the most advanced machining processes available today. It utilizes subtractive machining techniques that offer endless possibilities for part creation. The technology uses cutting tools that work on 5 axes to cut the workpiece into the desired shape and size.view Cast Iron vs Steel: Which One to Choose for Your CNC Project?January 15, 2024Cast iron vs steel is one of the many comparisons common among manufacturers. What are the differences between them? Read on and you can get the answer.view
Cast Iron vs Steel: Which One to Choose for Your CNC Project?January 15, 2024Cast iron vs steel is one of the many comparisons common among manufacturers. What are the differences between them? Read on and you can get the answer.view Introduction to MetalworkingOctober 24, 2023Humans have developed many methods of metal fabrication, which is the processing of metal into various shape types. Here we will introduce some of the concepts and processes of this metal working method.view
Introduction to MetalworkingOctober 24, 2023Humans have developed many methods of metal fabrication, which is the processing of metal into various shape types. Here we will introduce some of the concepts and processes of this metal working method.view Brass vs Bronze vs Pure Copper vs Copper Alloys: An in-depth look at their differencesSeptember 26, 2023The comparison of brass to bronze to copper and its copper alloys is crucial as these metals can be easily confused. In addition, the distinction between these materials is also very subtle. As a result, development engineers or designers can be seriously confused when it comes to material selection. In particular, the similarity of these metal constituent elements makes it more difficult to classify and select them.view
Brass vs Bronze vs Pure Copper vs Copper Alloys: An in-depth look at their differencesSeptember 26, 2023The comparison of brass to bronze to copper and its copper alloys is crucial as these metals can be easily confused. In addition, the distinction between these materials is also very subtle. As a result, development engineers or designers can be seriously confused when it comes to material selection. In particular, the similarity of these metal constituent elements makes it more difficult to classify and select them.view Chip Types Of Automatic Gear Production Line And Application Scope Of Chip Treatment SystemApril 12, 2022This paper studies the characteristics of different kinds of chips produced in the process of gear machining and gives the corresponding treatment scheme, which provides technical support for the auto...view
Chip Types Of Automatic Gear Production Line And Application Scope Of Chip Treatment SystemApril 12, 2022This paper studies the characteristics of different kinds of chips produced in the process of gear machining and gives the corresponding treatment scheme, which provides technical support for the auto...view