When designing parts for plastic injection molding, there is an important but often overlooked material consideration. It involves the glass transition temperature, or Tg. We'll get into the underlying science later, but for now, keep in mind that there is a temperature at which amorphous materials transition from a glassy/rigid state to a leathery/rubbery state.
The glass transition temperature is the temperature at which an amorphous polymer transitions from a hard/glassy state to a soft/leathery state, or vice versa. The Tg is directly related to the strength and potential uses of a material in a particular end-use application. The glass transition temperature is linked to the mechanical properties of a polymer. These include tensile strength, impact strength, modulus of elasticity, and operating temperature range, as shown in Figure 1.
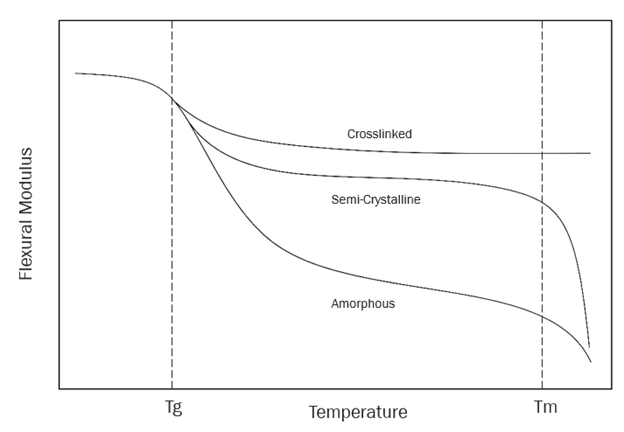
Figure 1: Flexural modulus vs. temperature.
There are two classes of polymers: Thermoplastics and Thermosets. Thermoplastic polymers are further divided into two groups: amorphous, such as polycarbonate (PC) and polystyrene (PS), and semi-crystalline, such as polypropylene or acetal.
To take a closer look here, let's take a digression back to high school chemistry class. Don't worry, it won't take long. We'll start on the first day when the teacher asks the question, "What are polymers?" The smart guy sitting up front studying ancient languages for fun knows that "poly" and "mer" are the Greek words for "many parts," so he concludes, "Polymers are long chains of smaller molecules joined together by a process called polymerization, with molecular weights in the hundreds to hundreds of thousands."
| Thermoplastic | Thermosetting | ||
|---|---|---|---|
| Amorph | Kristallin | ||
| Chain structure | Random/Unordered | Ordered/Stable | Networked |
| Melting point | Not defined/slow softening | Clear/crystalline decomposition | No Schelz point |
| Shrinkage rate | Low | High | Low |
| Appearance | Transparent | Opaque | Varies |
| Chemical resistance | Low | High | High |
| Examples | ABS, PC, PS | PP, PET, POM | Epoxid, LSR |
What is meant by molecular weight? More importantly, who cares? It should interest anyone who designs plastic parts. The molecular weight of a polymer determines the length of the "long chains" just mentioned, and thus its physical properties. For example, while a hydrogen molecule weighs only 1.01 g/mol (molar mass) and a carbon molecule weighs 12.01 g/mol, a single molecule of high-density polyethylene (HDPE) - which is nothing more than a chain of these two molecules - can weigh 250,000 g/mol or more.
Whether it's polyethylene terephthalate (PET) molecules weighing 8000 to 31,000 g/mol or polystyrene (PS) molecules weighing 400,000 g/mol, these centipede-like chains of monomers, which are the building blocks of polymers, arrange themselves into either amorphous or semi-crystalline structures.
Amorphous polymers have a randomly arranged/disordered chain structure. Below the glass transition temperature, they are hard and brittle. When heated, they slowly soften and become leathery/rubbery. This transition is the glass transition. When further heat is applied, they gradually melt (become moldable) after exceeding Tg to a temperature where the polymer begins to flow viscously. Common examples of amorphous polymers are hard, rigid materials such as polystyrene (PS) and polymethyl methacrylate (PMMA), which are used in their glassy state and well below their glass transition temperature.
Semi-crystalline polymers have highly ordered crystalline regions in addition to amorphous regions. The amorphous regions exhibit the properties just described. In semi-crystalline materials, on the other hand, the crystalline regions remain highly ordered after the glass transition temperature (Tg) is exceeded and form the structure of the material. For this reason, many semi-crystalline materials can be used above their Tg. Semi-crystalline materials such as polypropylene (PP), which has a Tg of about -20 °C, are used above their Tg in applications such as garden furniture, which is robust and flexible in the warm summer months but can become brittle in the cold winters.
Thermoplastic polymers have cross-links that bind their chains together. These bonds form between the chains, forming one large molecule. Think about this the next time you pick up a bowling ball. Cross-linking creates a robust chain structure, allowing elastomeric materials, such as silicone, to be used well above their Tg. Other thermoplastic materials are typically used below their Tg and are quite rigid, such as phenols. The cross-links form such strong bonds between molecular chains that the melting point of thermoset materials is above their decomposition temperature.
Tg for Common Molded Plastics
| Material | Tg in degrees Celsius |
| GPPS - Universal polystyrene | 100 |
| HDPE - High Density Polyethylene | -120 |
| LCP - Liquid Crystal Polymer | 120 |
| LSR - liquid silicone | -125 |
| PC - polycarbonate | 145 |
| PEEK - Polyetheretherketone | 140 |
| PEI - Polyetherimide | 210 |
| PMMA - polymethyl methacrylate | 90 |
| PP - Polypropylene (atactic) | -20 |
| PPS - Polyphenylene sulfone | 90 |
| PSU - Polysulfone | 190 |
| SPS - Syndiotactic Polystyrene | 100 |
Amorphous polymers are often transparent (polycarbonate and acrylic are two examples) rather than opaque, as are most semi-crystalline materials. They generally have better dimensional stability and are less likely to warp during the molding process. They are generally resistant to hot water and steam (think plumbing materials) and have good stiffness and impact resistance. As mentioned earlier, they tend to soften gradually when exposed to heat.
Partially crystalline thermoplastics have very strong molecular bonds due to their internal structure. This property makes them resistant to aggressive chemicals. Like Teflon, many of them offer a low coefficient of friction, making them a good choice for bearing and wear surfaces or where there is a heavy structural load. They are also much more resistant to fatigue than amorphous polymers. They soften when exposed to heat, but can be used above their Tg because the crystalline regions retain their structure up to the melting temperature of the polymer.
With their cross-linked internal structure, thermoset materials have very good chemical resistance, dimensional stability and heat resistance. Thermoplastic materials vary from transparent to opaque. They can be elastomeric or rigid. They can also be used above and below their Tg and have no melting point.
There is no doubt that we are dealing with a complex subject here. We hope that these technical remarks have helped to bring a little more clarity to this subject area.
Are you working on an precise machining project? Contact us at +86-0755-28025755 or sales@richconn.com.cn. We are always happy to talk about polymers.
 CNC Cutting Aluminum Sheets for Electronics EnclosuresFebruary 29, 2024Elevating Electronics Enclosures with CNC PrecisionIn today's fast-paced technological landscape, where precision and efficiency are critical, the manufacturing of electronics enclosures demands i...view
CNC Cutting Aluminum Sheets for Electronics EnclosuresFebruary 29, 2024Elevating Electronics Enclosures with CNC PrecisionIn today's fast-paced technological landscape, where precision and efficiency are critical, the manufacturing of electronics enclosures demands i...view Some Knowledge You Need to Learn About Tapped HoleNovember 16, 2023Do you need taped holes? This article covers the factors you must consider to manufacture the tapped hole during the process.view
Some Knowledge You Need to Learn About Tapped HoleNovember 16, 2023Do you need taped holes? This article covers the factors you must consider to manufacture the tapped hole during the process.view The Basics of Engineering Drawing and Tips for NewcomersApril 7, 2024The article gives some useful tips on how to make engineering drawings easily to help you better communicate your ideas clearly.view
The Basics of Engineering Drawing and Tips for NewcomersApril 7, 2024The article gives some useful tips on how to make engineering drawings easily to help you better communicate your ideas clearly.view What Is a Shaft?October 30, 2023A shaft is basically a rotating part of any machine, having a circular cross-section, which is used to transmit power from one part to another or from a power generating machine to a power absorbing machine. To transmit power, one end of the shaft is connected to the power source and the other end is connected to the machine. Shafts can be solid or hollow as per requirement, hollow shafts help in reducing weight and provide advantages.view
What Is a Shaft?October 30, 2023A shaft is basically a rotating part of any machine, having a circular cross-section, which is used to transmit power from one part to another or from a power generating machine to a power absorbing machine. To transmit power, one end of the shaft is connected to the power source and the other end is connected to the machine. Shafts can be solid or hollow as per requirement, hollow shafts help in reducing weight and provide advantages.view 7 Best Tips for Sandblasting (The Complete Basics to Get Started)August 24, 2023Sandblast is a process that has been used for many years. It is used to clean, etch, and prepare surfaces before they are painted or coated with another material. Unfortunately, sandblasters are machi...view
7 Best Tips for Sandblasting (The Complete Basics to Get Started)August 24, 2023Sandblast is a process that has been used for many years. It is used to clean, etch, and prepare surfaces before they are painted or coated with another material. Unfortunately, sandblasters are machi...view Top 12 CNC Machining Manufacturers in ChinaJune 6, 2024Among the numerous CNC machining services, why choose CNC machining manufacturers in China for your projects? This article will reveal the answer for you.view
Top 12 CNC Machining Manufacturers in ChinaJune 6, 2024Among the numerous CNC machining services, why choose CNC machining manufacturers in China for your projects? This article will reveal the answer for you.view
 EN
EN
 ru
ru 


